Trabexus®
Trabecular Osteoinductive Biocement‡
Self-setting calcium phosphate matrix with engineered allograft bone particles for optimized compressive strength and resorptive characteristics.
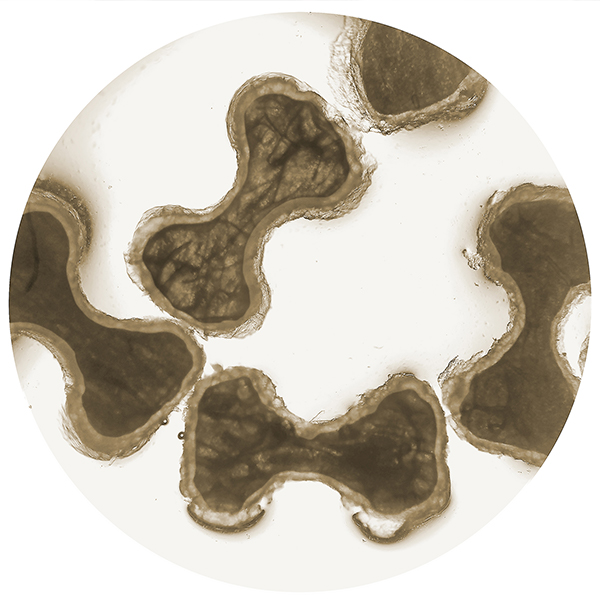
Trabs® viewed under high magnification, partially demineralized layer is clearly visible on outer surface.

Trabs® prior to mixing with calcium phosphate
Product Overview
The Optimal Bone Void Filler
The cornerstone of Trabexus is the thoughtful design of the allograft component. In Trabexus, the allograft component is formed into “hourglass” shapes (known as Trabs) using a proprietary manufacturing process. This novel shape provides multiple benefits, including increased “interconnectivity” of adjacent particles, creation of channels for accelerated remodeling and greater overall surface area. These factors optimize the ratio of compressive strength to allograft content. The Trabs are partially demineralized, exposing key osteoinductive proteins, while preserving a core of mineralized cortical bone allograft, which further optimizes the product’s resorption characteristics.
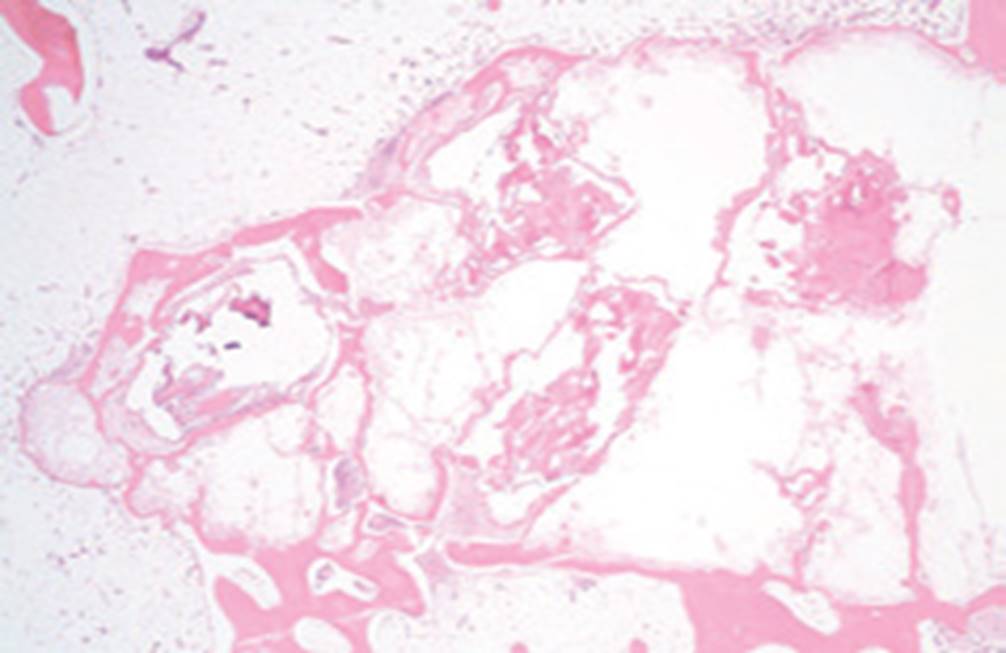
Trabexus
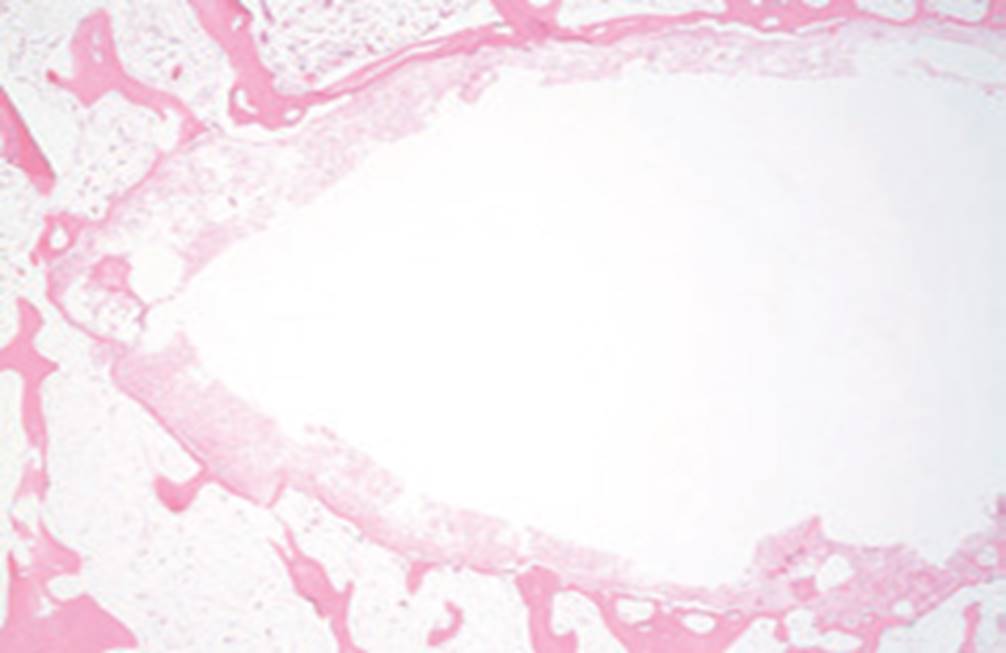
Standard Cement
Surgically created bone defects in a rabbit model were filled with Trabexus and compared to defects filled with standard calcium phosphate cement.
8 weeks post surgery, Trabexus filled defects demonstrate signs of cell infiltration and remodeling.
| Composition | Resorbable calcium phosphate matrix with partially demineralized allograft particles “Trabs” |
|---|---|
| Compressive Strength | 25MPa |
| Osteoconductive | Material crystallizes into hydroxyapatite upon setting, the mineral phase of native human bone |
| Osteoinductive‡ | Allograft component is partially demineralized, exposing key osteoinductive proteins |
| Resorption Profile | More rapid compared to calcium phosphate cements |
| Thermal stability | Does not require refrigeration prior to use |
| Resistant to lavage | Solidifies in a wet field |
| Sizes | 3cc, 5cc, 10cc |
†Trabexus is covered by United States patents 9,072,720 and 10,046,090.
‡Allograft component demonstrated osteoinductivity in athymic mouse model submitted for 510(k). Refer to 510(k) summary K143547. Data on file at Vivorté, Inc.
